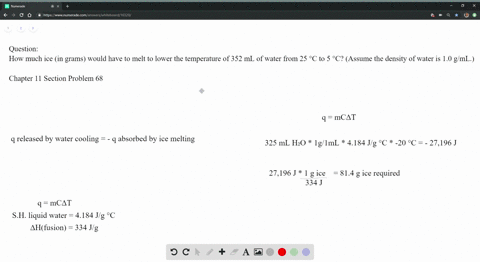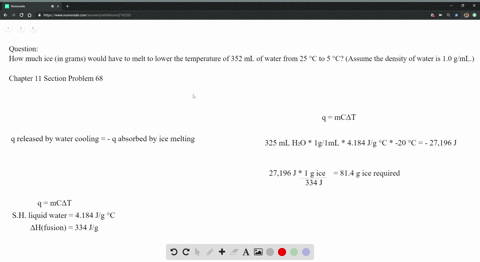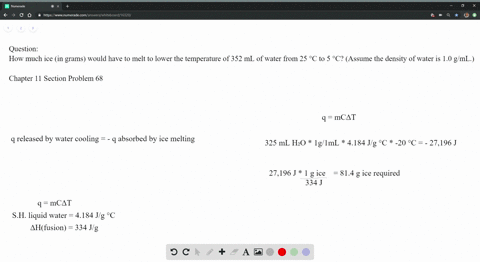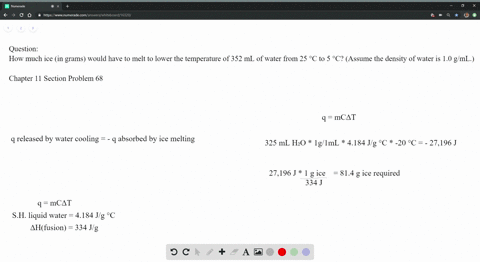
The strong intermolecular force also means that the solid maintains a fixed volume and shape (all molecules are held tightly in place). The strong forces of attraction prevent the molecules from moving, so they vibrate around a fixed point. They are also arranged regularly, or in other words, arranged in a lattice, as this minimises the distance between particles. 
This causes their molecules to be tightly bound (solids have the smallest distance between molecules). Solids have very strong intermolecular forces of attraction. Relate the properties of solids, liquids and gases to the forces and distances between molecules and to the motion of the molecules.Gas molecules are far apart and randomly arranged. The molecules in a liquid are close together and are irregularly arranged. The molecules in a solid are close together and arranged in a regular pattern. Describe qualitatively the molecular structure of solids, liquids and gases.We apologise for the inconvenience, but hope that the new images will provide you with an even better learning experience. Disclaimer: Due to unforeseen difficulties, we have had to take down the images on this notes page.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
